Efflorescence in concrete units is usually observed as a white deposit on the surface. This deposit can occur as a slight haze up to a crusty layer. Efflorescence is not a structural problem, it does not affect concrete strength or durability. But it is unsightly and lowers the perceived value of the concrete.
Efflorescence is usually composed of salts that are deposited on a concrete surface. It’s worth noting that any concrete product that contains cement has the potential to produce efflorescence. There are four factors that combine to produce efflorescence, and they can occur during production, or once the units are installed on the jobsite. In this article, we will focus on what concrete producers can do to limit efflorescence.
As you know, there’s a lot of chemistry involved in concrete, so we have to get into some chemical reactions to understand what’s going on. When water is added to Portland cement in the mixer, a chemical reaction called hydration occurs. Cement hydration is what causes concrete to harden and gain strength, so it’s very important we understand hydration and maximize it.
The hydration reaction between cement and water forms calcium silicate hydrate (CSH) or “good” gel. This is the glue that hardens and holds concrete together. Unfortunately, the reaction is not very efficient, and a by-product called calcium hydroxide, a “bad” gel, is also formed.
One way to limit calcium hydroxide is to use pozzolans such as fly ash or slag to replace some of the cement. Fly ash and slag are able to react with calcium hydroxide, and this reaction produces more CSH gel, which is what we want. Efflorescence can also result from other soluble salts, called alkalies, usually found in the aggregates.
During mixing: It is very important for concrete strength and durability that there is sufficient water available during mixing to hydrate the cement particles properly. Therefore, maximizing water during mixing is highly desirable. Adding as much water as possible short of pulling, picking or slumping during mixing will lead to products with higher strength and lower absorption.
After mixing: Concern over excessive water in the units comes into play after mixing, when the concrete products reach the curing stage - in the kiln or out in the yard.
Once the units are in the yard, rain and condensation can penetrate into the units, creating excess moisture conditions. Units that are exposed to repeated cycles of wetting and drying, such as during a wet spring or fall, may have increased potential for efflorescence.
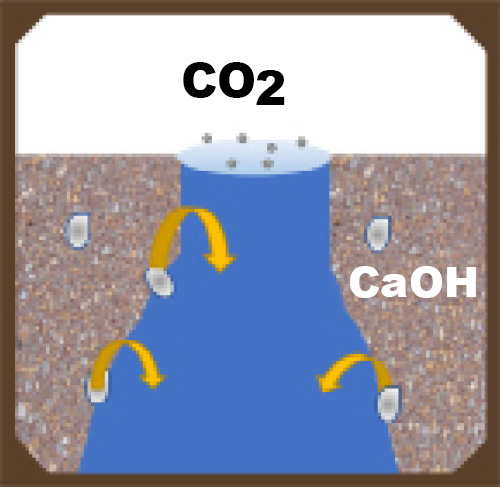
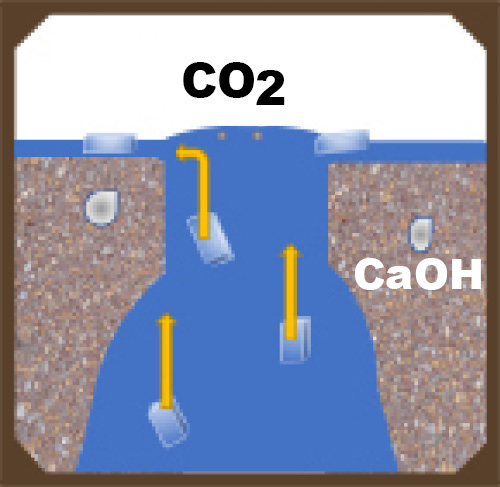
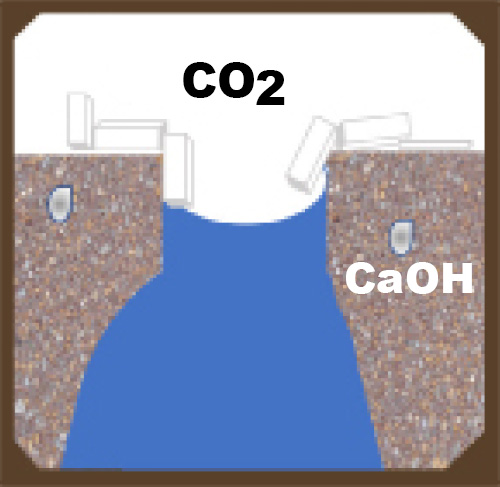
All manufactured concrete products have a network of interconnected voids. Connected voids serve as pathways for moisture migration into and out of the concrete unit. Voids can be minimized but not eliminated. Even if you have the best mix design, materials, equipment and compaction possible, there will still be voids and pathways for salt solutions to migrate to the surface.
These pathways can be reduced by making the concrete more dense, and thus less able to absorb water. Admixtures, including water repellents and densifiers, can aid in limiting water penetration.
When two things occur in different conditions (for example one hot and one cold), the environment will try to find balance between the two situations, and will react to reach the middle or equilibrium. This is a driving force. Driving forces will try to balance humidity and temperature in a concrete unit with ambient conditions. This may cause moisture in the unit that contains the calcium hydroxide salt solution to move to the surface of the concrete.
During curing and storage of concrete units, many common practices can cause a driving force to occur. These include: